Blog
You don’t need to spend countless hours perusing your typical online sources when you have this! Stay informed today by becoming an AARS member and receiving the Hot Topics!

The American Acne and Rosacea Society (AARS) Highlights #RosaceaFacts During Rosacea Awareness Month
April is Rosacea Awareness Month, and the American Acne and Rosacea Society (AARS) continues to...

FDA Accepts NDA from Journey Medical for Modified Release Minocycline
The FDA has accepted the New Drug Application (NDA) from Journey Medical Corp. for DFD-29...

Response Statement from the American Acne & Rosacea Society to the Valisure Citizen Petition on Benzene in Benzoyl Peroxide Drug Products
On March 5, 2024, Valisure, an independent analytical laboratory located in New Haven,...
New Research on Inflammation and Transposons Adds to Understanding of Antibiotic Resistance
Having found a novel genetic arrangement that may help Bacteroides fragilis protect itself from...

AARS Addresses Psychosocial Impact of Acne on World Teen Mental Wellness Day
In recognition of World Teen Mental Wellness Day on March 2, members of the American Acne and...

AARS Is Now Accepting Applications for Research Grant Awards
The American Acne and Rosacea Society (AARS) is pleased to announce that it is now accepting grant...
Could Engineered C. acnes Treat Acne?
Researchers have validated in skin cell lines and in mice an engineered strain of C. acnes that...

Drug Prices in US Substantially Higher Than in Other Nations
Brand-name drug prices in the US are 4.22 times higher, on average, than those in 33 other...
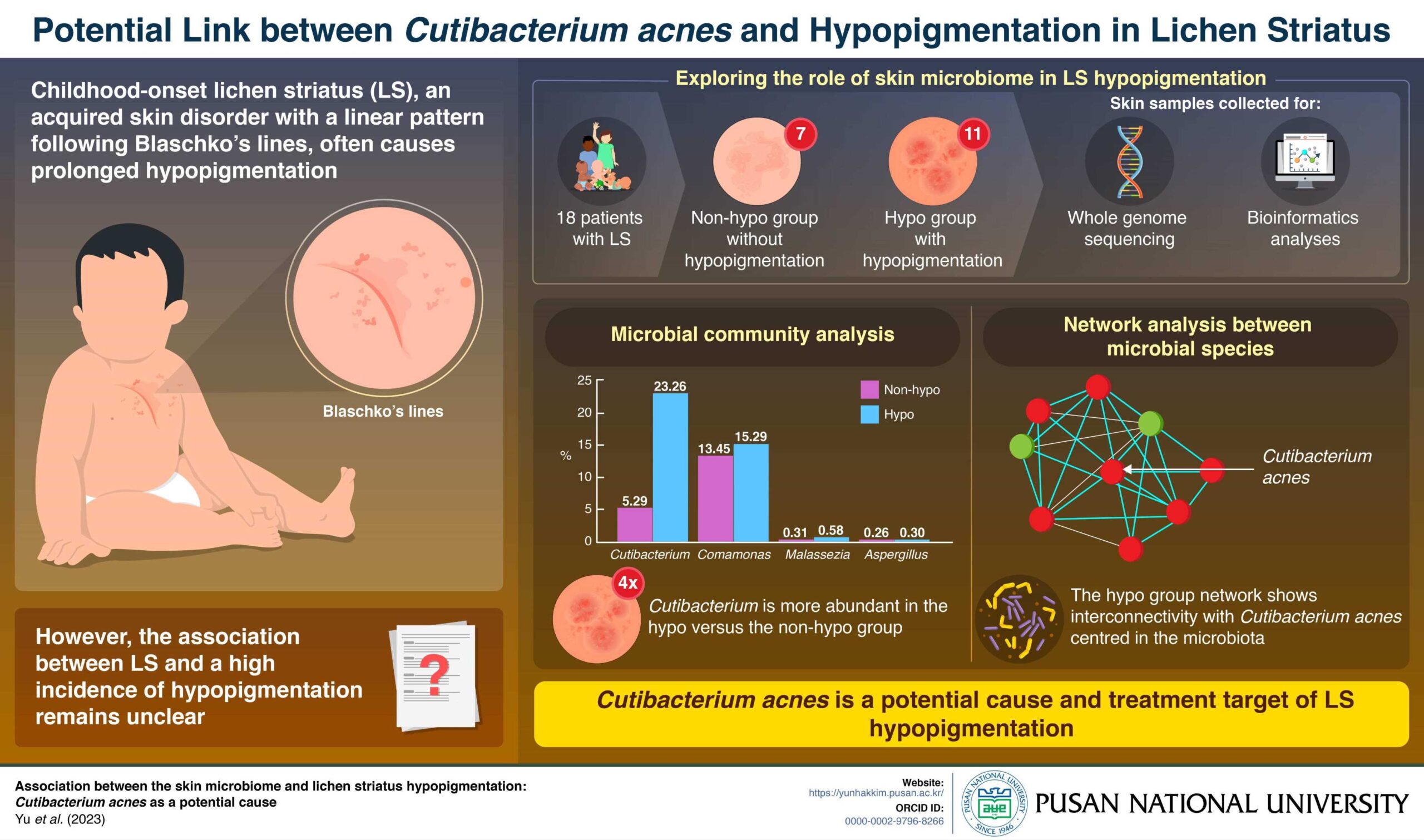
C. acnes May Be Associated with Hypopigmentation in Lichen Striatus
Cutibacterium acnes may be associated with the development of lichen striatus (LS)-associated...

New Research at Poultry Farms Reveals Pathway for Spread of Antimicrobial Resistance
Livestock farming has long been recognized as contributing to the development of drug resistant...
